Obtain corrosion rate versus time automatically with the Corrosimetry technique!

Introduction
The determination of the corrosion rate provides information about the behavior of a system such as the impact of an environment over a material, or the protective ability of an electrodeposited coating for example. Polarization resistance techniques are widely used to determine the general corrosion rate of the studied electrochemical system. The Linear Polarization Resistance (LPR) technique is commonly used as a standard (ASTM G 59).
Following the corrosion rate with a potentiostat for a long period of time may help understand evolutions occurring at the electrolyte/material interface. Usually, this investigation may turn out to be laborious due to the repeated collection of data and the repeated analysis.
BioLogic’s single-channel or multichannel potentiostats / galvanostats do the measurements and the analysis automatically thanks to the EC-Lab® “corrosimetry” technique which is described below.
Polarization resistance
The polarization of an electrochemical system consists of applying a voltage that is different from the equilibrium voltage of the cell. Depending on the polarization direction, it is referred to as anodic or cathodic polarization.
Using the Tafel theory, the polarization of the system (the overpotential) can be related to the current in the anodic area ηa=log 10 βalog(i/i0) and in the cathodic area ηc=−log 10 βc log(i/i0).
Corrosimetry (Rp vs. time)
The corrosimetry technique is available in the corrosion techniques section of EC-Lab®. It is based on the Linear Polarization Resistance (LPR) technique, which permits to determine the polarization resistance. The Tafel parameters, βaβa and βcβc, must be determined beforehand thanks to Tafel, VASP or CASP techniques and fill in the corrosimetry “Parameter Settings” window (Figure 1 below).
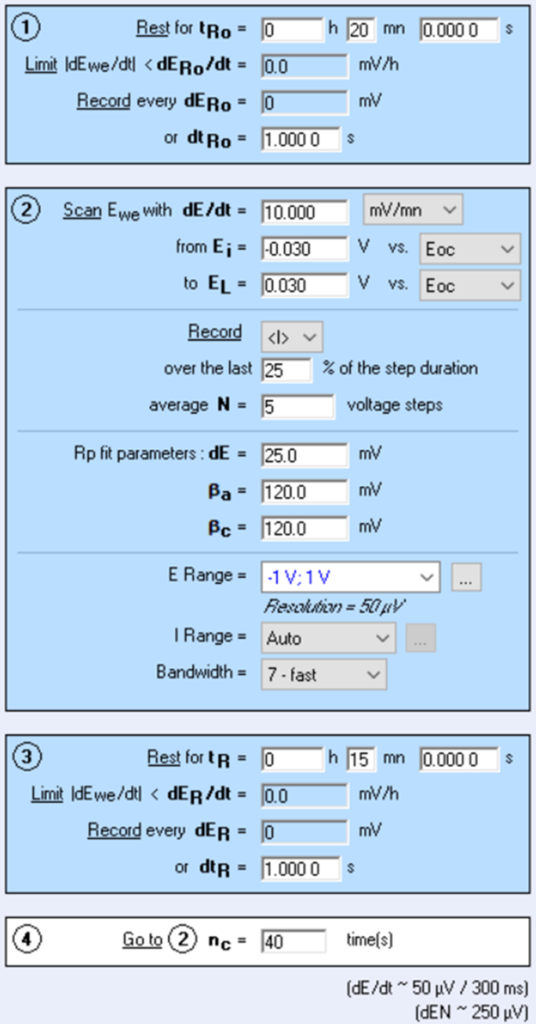
Figure 1: Corrosimetry “Parameter Settings” window
Please click on ‘Request Application Note’ and we will send you the full application note: ‘Obtain corrosion rate versus time automatically with the Corrosimetry technique!’.