Fourier transform infrared spectroscopy and imaging are valuable techniques for examining the chemical composition of biological materials by probing their vibration motions on the microscopic scale. Measuring chemical composition on the nanoscale can be achieved by AFM-IR techniques such as; photo-induced force microscopy (PiFM). In this application note nanoscale IR identification on biological tissue is shown and compared with X-ray absorption microscopy.
For this experiment fixed human skin samples were penetrated by topically addressing dexamethasone, a synthetic glucocorticoid comparable to cortisol. The skin samples were sliced into sections of 300nm thick which were cross-sectioned and deposited on a Si3N4 window afterwards (for X-ray absorption microscopy). The same area was then subsequently analyzed by PiFM.
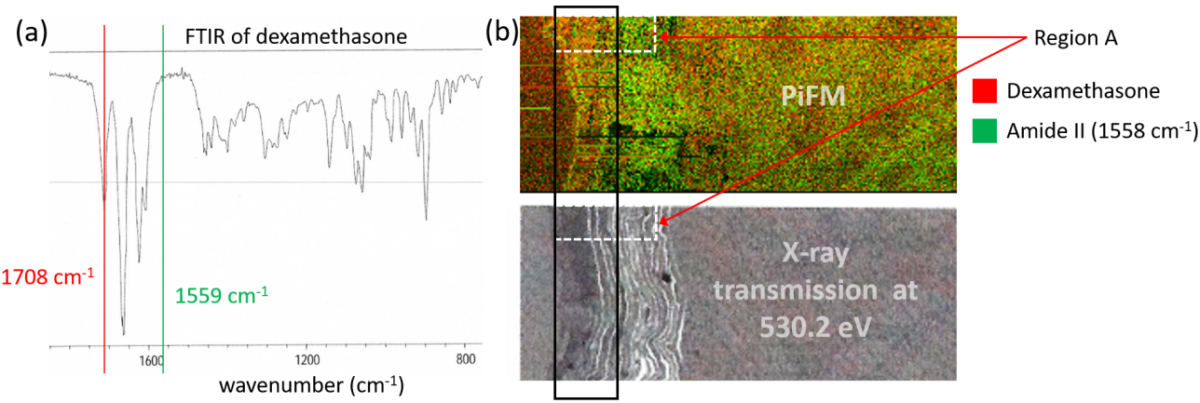
Figure 1. (a) FTIR spectrum of dexamethasone; (b) combined PiFM images (top) acquired at 1708 cm-1 for dexamethasone (red) and at 1558 cm-1 for corneocytes (green) and X-ray transmission image of the same region. Region A is further investigated by PiFM in Figure 2.
Given the FTIR spectrum of dexamethasone (Fig. 1a), PiFM images were acquired at 1708 cm-1 and 1559 cm-1 to highlight dexamethasone molecules and corneocytes (via amide II band), which are the cells in the outermost layer of the skin (stratum corneum). The amide I band was avoided since dexamethasone has a strong absorption at around 1660 cm-1.
PiFM images for dexamethasone (red) and corneocytes (green) are combined to map the two different components; the resulting chemical map is shown in Fig. 1b (top) and compared to the X-ray transmission image (below) at 530.2 eV, the maximum absorption energy for dexamethasone. From both PiFM and X-ray transmission images, we can see that the drug is primarily concentrated toward the top region of the stratum corneum, highlighted by the black rectangular box. This result is similar to previous work on X-ray microscopy [REF: K. Yamamoto et al., Eur. J. Pharm. Biopharm. 18, 30 (2017)].

Figure 2. Detailed view on the stratum corneum of fixed human skin: (a) AFM topography visualizing the structure of corneocytes; (b) chemical PiFM contrast related to amides probed at 1559 cm-1; (c) PiFM contrast related to dexamethasone at 1708 cm-1; (d) two PiFM images combined to map chemical components. Figures (a’)-(d’) show an enlarged view of the regions indicated by white boxes in (a)-(d).
Figure 2 shows PiFM images at higher magnification along with the topography. The top images (12×12µm) show the stratified structure of the stratum corneum; the bottom portion of these images coincides with the region A defined in Fig. 1b.
Fig. 2c shows how the drug is concentrated in the uppermost layer of the stratum corneum like seen in previous images. The bottom row of images (3×3µm) zoom into the indicated region in the top images (white box).
Figure 2b’ shows that no amide II signals are seen in between the corneocytes, most likely due to the intercellular lipid layer embedding the corneocytes. However, Fig. 2c’ shows that the topically applied drug penetrates into these lipid lamellae between the corneocytes and fills them partially; this is even more clearly visible in Fig. 2d’ (combined PiFM image, red for dexamethasone and green for corneocytes), which is magnified by 2X for easier observation.
With its spatial resolution that goes beyond X-ray microscopy and the lack of concern for radiation damage on soft tissue samples, PiFM is an attractive technique to acquire simultaneous chemical and morphological information in true nanoscale.
Credit: The samples were provided by E. Rühl, Freie Universität Berlin (Germany)
